| Toxys announces a €2 million funding round from KIKK Capital, InnovationQuarter, Zeeland Investment fund, Toxys management and Rabobank. This investment is being made to advance Toxys’ ambition to become the global leading supplier of expert mechanistic in vitro chemical safety testing services. These services provide animal-free safety testing of novel medicines, chemicals and consumer products. The patented ToxTracker® assay can identify toxic and carcinogenic properties of novel pharmaceuticals, agrochemicals and fine chemicals already at an early stage of drug/product development. ToxTracker can prevent the unnecessary rejections of highly valuable drug candidates and products, reduce drug/product development time lines and can prevent unnecessary animal testing. ToxTracker has the unique ability to identify the mode-of-action of toxic compounds which significantly improves the ability to predict and reduce risks for human health. ToxTracker is currently under review by the international authorities for official acceptance and inclusion of the test systems within the standard regulatory safety test strategy. Furthermore, with its soon-to-be launched novel ReproTracker® test, Toxys aims to fill a large unmet meet for an in vitro embryo toxicity test. With this new financial investment, Toxys is enabled to accelerate the growth of the company by expanding its operations in Europe and the United States. Toxys will also further broaden its portfolio of unique in vitro toxicology assays, speed-up international growth and work in acceptance and implementation of its toxicology assays for regulatory applications. Giel Hendriks, CEO of Toxys, commented: “We have made important progress on development of innovative animal-free tests for drug and chemical safety testing. I am very proud of the strong and dedicated Toxys team to execute our plans to become a trusted expert in toxicological research. With this new investment, we are ready to further expand our business globally and continue to work on the development of novel solutions for toxicological research”. Johan Sebregts, Investment manager KIKK Capital: “As a board member I have seen Toxys grow to a successful and profitable biotech company with a growing global client base. Now the first product of Toxys, ToxTracker, has proven itself in the pharmaceutical and chemical industry it’s time for further international expansion and the market introduction of some exiting new products. KIKK Capital is looking forward to support the developments of Toxys and is convinced that with the investment Toxys will be able to realize its full international potential”. More information: www.toxys.com |
 At 21 september Toxys was awarded a €50.000 grant for a head-start project to further develop the ReproTracker. Toxys is dedicated to develop innovative in vitro toxicity assays that provide insight into the mechanisms of toxicity. Toxys’ lead product ToxTracker is a genotoxicity assay that provides mechanistic insight into genotoxicity and is available on the market. Toxys has now applied their knowledge and expertise to validate a novel assay, ReproTracker, which provides mechanistic insight into reproductive toxicity. About ReproTracker ReproTracker is an in vitro assay to visualise the key events during early embryonic development and thereby reliably assess the potential developmental toxicity hazards of new drugs and products. ReproTracker meets the strong demand from industry for better in vitro tests to predict human health hazards of their products during the early phases of product development. The assay is build on the technical and scientific background that has been developed by Toxys. We will combine our expertise on stem cell differentiation with the reporter technology that was applied in our ToxTracker assay and generate the ReproTracker reporter system that can identify developmental toxicity upon exposure to novel drugs, chemicals and materials. About EIT Health Toxys was awarded the head-start project from EIT Health, which is one of the largest healthcare initiatives worldwide. Its goal is to sustainably advance the foundations of healthcare and thus promote the future conditions for healthier living and wellbeing of people across Europe. EIT Health is leveraging the expertise of more than 130 leading organisations spanning key areas of healthcare such as Pharma, MedTech, Payers, Research Institutions and Universities. Chosen by the European Institute of Innovation and Technology (EIT) to form EIT Health, the consortium offers best-in-class research capabilities, higher education and business expertise. With a budget of 2 billion EUR over the next decade, it will purposefully invest in Europe’s best entrepreneurial talents and creative minds to foster the development and commercialisation of smart product and service solutions in the health sector, addressing the challenges imposed by demographic change and ageing societies. More information about Toxys. 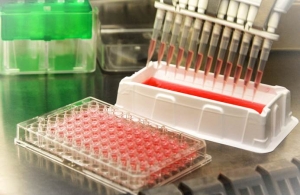 Industries are continuously developing new types of substances that are used in the manufacturing of new products, such as medicines, cosmetics and food. These substances should be thoroughly tested for safety before they can be marketed. Industry, however, is experiencing limitations with the tests that are required in the legislation, and it happens far too frequently that a substance is wrongly classified as being either safe or harmful. As a result of these erroneous tests, unnecessary investments are made in harmful substances, and the development of valuable drug candidates is terminated improperly. The innovative biotech company Toxys from Leiden has solved this problem by developing a much better test, the ToxTracker assay. ToxTracker is a unique test, based on stem cell technology, which is able to very quickly and reliably identify the main cancer-causing properties of new materials. The substance to be tested is dripped onto the test plate (assay), developed by Toxys. Following this, there is a reaction, and data is collected and interpreted. The test is more sensitive and specific than the tests that are currently available. The Toxys test reduces the percentage of current in vitro false test results from 30% to 3%. "We obviously see to what extent they are toxic, but what is really unique is that we provide mechanistic insight into the various harmful properties of materials," says Dr. Giel Hendriks, the founder of Toxys. "By understanding how substances react in our cells, we can determine much more reliably if they are possibly carcinogenic or not." Great benefits for industry It is of great importance for companies that the materials and ingredients they use have no harmful effects on human health. If new substances or ingredients are to be put into the products, companies want to know as soon as possible whether it is worthwhile to invest further in the development of a substance, or if for health reasons it is not suitable. Significant adverse effects may result if the toxicity only emerges once the product is being used, which may lead to health problems, damage claims, and loss of reputation. Another advantage of the ToxTracker technique is that fewer test animals are needed. Pharmaceutical and chemical substances with the strongest carcinogenic properties are eliminated, and therefore do not need to be tested on animals. Investment ZIF & InnovationQuarter Toxys is using the investment to bring the existing products to the markets, to accelerate the development of new tests, and to move to a private laboratory. "With this investment Toxys can work on new innovative tests and services, and thereby contribute to the development of better drugs and safer products," says Giel Hendriks. Johan Sebregts, Fund Manager of ZIF: "From the first meeting on, we were impressed with the performance of Toxys. The validated test, the involvement of LUMC and Leiden University, and the fact that there are various reputable clients gave us immediate confidence. This, coupled with new product developments and the growing market for early stage testing, ensures that we have great confidence in the potential of Toxys. Through the investment of ZIF and InnovationQuarter, and the contribution of our knowledge and experience, we want to contribute to the realization of Toxys’s growth." Toxys is a spin-off from the Department of Human Genetics at Leiden University Medical Centre (LUMC) and was founded in 2014 by scientist Giel Hendriks. More information about Toxys. |
ARCHIVES
March 2020
CATEGORIES
All
|

